Cost Leadership with
Innovative Technology
Commercial-scale
Suzhou Base
State-of-the-art facility for GMP manufacturing to support:
Global clinical trials
Partnership programs
Scaling of manufacturing
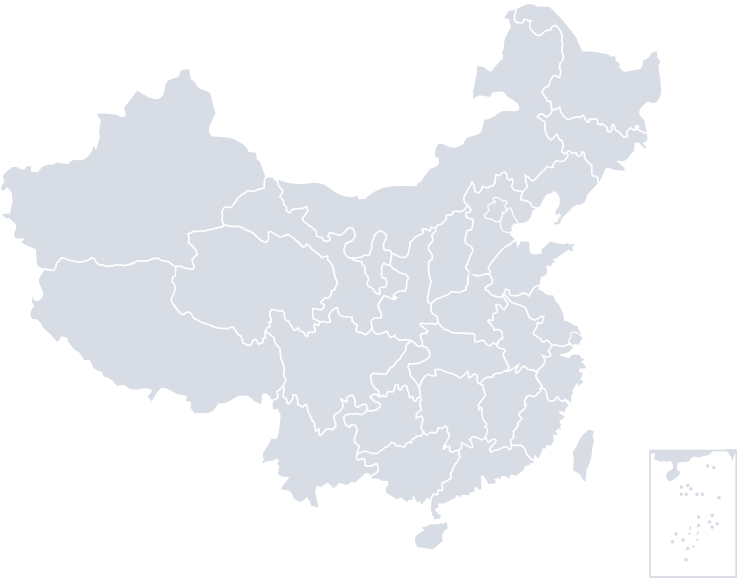

Wuzhong, Suzhou